Sakshi Duklan; Supriyo Saha; Vikash Jakhmola; Nidhi Gairola; Pallavi Pandey; Mahipal Singh; Sarkar Mohammad Abe Kawsar
Abstract
A newer generation pyrimidine derivatives were designed, synthesized, and evaluated in vitro alpha amylase and bacterial growth inhibitor. The molecules' design fully depended upon ...
Read More
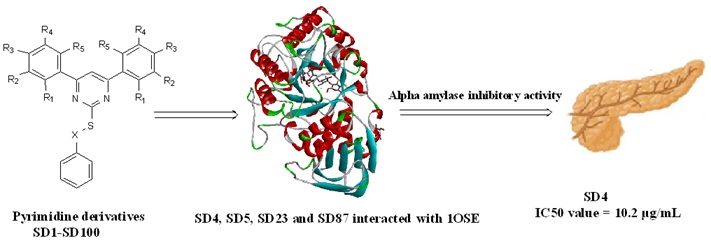
A newer generation pyrimidine derivatives were designed, synthesized, and evaluated in vitro alpha amylase and bacterial growth inhibitor. The molecules' design fully depended upon the structural features of previously pyrimidine derivatives. Then all the designed molecules (SD1-SD100) were docked with 1OSE pig pancreatic alpha-amylase isoenzyme. S-[4-(2-hydroxyphenyl)-6-phenylpyrimidin-2-yl] benzenecarbothioate, S-(4,6-diphenylpyrimidin-2-yl) benzenecarbothioate, S-[4-(4-hydroxy-3-methoxyphenyl)-6-phenylpyrimidin-2-yl] benzenecarbothioate, and S-[4,6-bis(4-hydroxyphenyl)pyrimidin-2-yl] benzenecarbothioate showed good docking interaction scores, as compared to acarbose. The interacting residues of the synthesized molecules and 1OSE showed similar amino acid lining as present in the active site. The synthetic procedure of the molecules was divided into two steps such as synthesis of chalcone derivative using aromatic aldehyde and acetophenone, reaction between chalcone and thiourea to form substituted pyrimidine-2-thiol, then finally substituted pyrimidine-2-thiol and benzoyl chloride reacted in presence of glacial acetic acid to obtain the best docked molecules. All the molecules show characteristic peaks in FTIR, 1H-NMR and Mass spectrometric data. Among all the synthesized molecules S-[4-(2-hydroxyphenyl)-6-phenylpyrimidin-2-yl] benzenecarbothioate showed best in vitro alpha amylase inhibition activity. Also, all synthesized molecules showed moderate to good antibacterial activities.